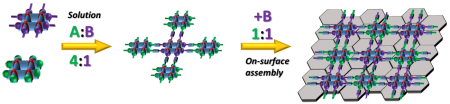|
One of our main topics deals with so-called Supramolecular grids. These are multicomponent organometallic complexes with very regular rectangular geometry. This feature predetermines them for use in nanoelectronics. In our group we want to take advantage of this spatial arrangement to study their properties on surface for potential applications in nanoelectronics. Another important feature of the grids is their metalo-responsive self-assembling formation, which we would like to use in creation of recyclable surface-bound platform for sensors and (electro)catalysis. Furthermore, the mentioned multicomponent nature means that with appropriate functionalizition these grids could be used as multivalent platforms for the studies of biochemical interactions.
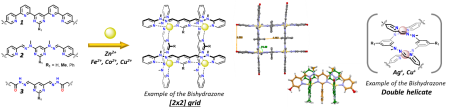
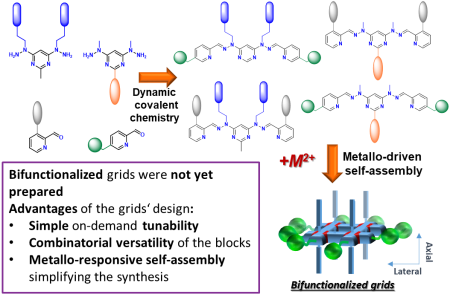
The work is based a series of peripherally decorated metallosupramolecular grids, which create extended interconnected 2D arrays, bioactive multivalent compounds and responsive materials. The development of novel and versatile (bi-)functionalized grids with accurately spatially distributed and tailored functionalities combined with a help of dynamic covalent chemistry (DCC), will allow to address all below presented topics with just a small library of easily accessible starting complexes.
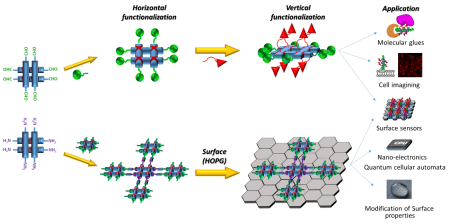
1) The multivalency studies: Newly designed multifunctional grids with different active moieties situated in two orthogonal planes will be prepared and used as a customable platform for analytic and biochemical use (e.g. Multivalent inhibitors, Cell imagining, Molecular glues).
2) The surface applications: Several ideas will be tested to achieve a stable surface deposition of herein prepared grids. Strategies, such as intensification of the surface coordination through addition of strongly interacting moieties (e.g. pyrenes) or through grids oligomerization (Figure 1, Bottom) as well as direct covalent attachment will be used to address different applications. Among others, the highly regular geometry of the grids array is an ideal frame for molecular electronics, spintronics and magnetics. Of particular appeal is the idea of “quantum cellular automata”[1], where information storage is based on an array of the different redox states rather than on points capable of current transmission.
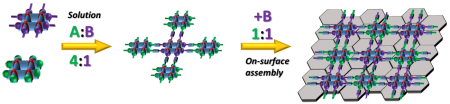
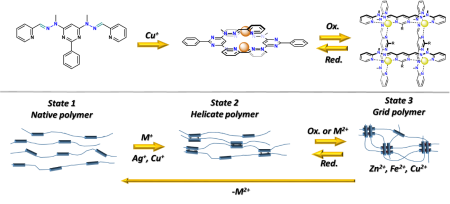
3) The material chemistry: The grid ligands will be incorporated into the polymer matrix and used to alter its physical properties through metal asserted control of different complexation states (ligand/helicate/grid). Furthermore, an application of the electrochemical oxidation/reduction should result in reversible switching between two states providing “smart material”, usable in, for example, artificial muscles.
- Ruben , J. Rojo , F. J. Romero-Salguero , L. H. Uppadine and J.-M. Lehn , Grid–Type Metal Ion Architectures: Functional Metallosupramolecular Arrays, Angew. Chem., Int. Ed., 2004, 43, 3644 —3662
- G. Hardy Metallosupramolecular grid complexes: towards nanostructured materials with high-tech applications, Chem. Soc. Rev., 2013, 42, 7881 —7899
- Holub, J.; Santoro, A.; Stadler, A.-M.; Lehn, J.-M. Peripherally Multi-Functionalised Metallosupramolecular Grids: Assembly, Decoration, Building Blocks for Dynamic Covalent Architectures, Inorg. Chem. Front. 2021, 8 (23), 5054-5064
|
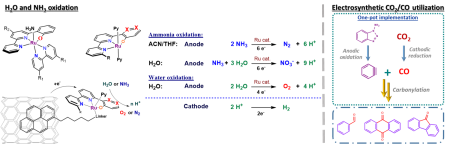
Even though, a lot of effort is invested into creation of evermore complicated molecules for drugs and materials, it is small molecules which have the power to change our world. For example, water oxidation is considered as a key for sustainable energy as green source of H2 and CO2 reduction and valorisation could help reduce our dependence on fossil fuels and thus slow down global warming.
In our group we are developing Ruthenium based organometallic (electro)catalysts for water oxidation and its newly emerging alternative ammonia oxidation. With robust catalysts in hand, in the future our focus will turn to other transformations such as CO2 and N2 reduction and/or utilization of cheap 1st row transition metals (e.g. Fe, Co, Mn).
- Matheu, R.; Garrido-Barros, P.; Gil-Sepulcre, M.; Ertem, M. Z.; Sala, X.; Gimbert-Suriñach, C.; Llobet, A. The Development of Molecular Water Oxidation Catalysts. Nat. Rev. Chem. 2019, 3 (5), 331– 341
- Dunn, P. L.; Cook, B. J.; Johnson, S. I.; Appel, A. M.; Bullock, R. M. Oxidation of Ammonia with Molecular Complexes. J. Am. Chem. Soc. 2020, 142 (42), 17845– 17858,
- Holub, J.; Vereshchuk, N.; Sánchez-Baygual, F.-J.; Gil-Sepulcre, M.; Benet-Buchholz, J.; and Llobet A. Synthesis, Structure, and Ammonia Oxidation Catalytic Activity of Ru-NH3 Complexes Containing Multidentate Polypyridyl Ligands Inorg. Chem. 2021, 60 (18), 13929–13940
|