Electrosynthesis approach to CO2 and CO capture for preparation of value-added chemicals
Electrochemical Reduction:
Beyond CO2/CO: Synthetic electrochemistry
Under the pressure of global warming and increasing level of CO2, much effort is being made to find a green transformation that can reduce its atmospheric levels. Most of the work is devoted to (electro)chemical reduction of CO2 into CO, HCOOH, CH4 etc. Few reports deal with direct CO2 electrochemical transformations such as carboxylation. Even fewer try to couple CO2 reduction in combination with another chemical transformation. The illustrative example is a report on the two-step CO2/CO reduction process combined with carbonylative Sonogashira or Suzuki couplings.[1] This work aims to couple two independent electrochemical processes in one classic organic reaction. More specifically, coupling a CO2/CO reduction with an oxidative electro-generation of aryne to achieve an arene carbonylation reaction for aldehyde, fluorenone or anthraquinone production. (Figure 1)
Methodology
1) Aryne electrogeneration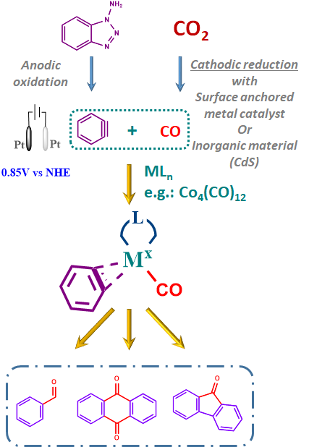
Arynes are reactive and useful intermediates in the synthesis of aromatic compounds; however, due to their reactivity, it is necessary to generate them in situ and react immediately. Many different methodologies are already relying on the stoichiometric use of particular reagents to generate an aryne triple bond. Especially interesting for the scope of this work is a recent study on the electrochemical generation of arynes from 1-aminobenzotriazole for stereoselective arylation of β‐Ketocarbonyls.[2] The protocol takes advantage of a standard electrochemical set-up and commercially available materials providing a solid starting point for further build-up. (Figure 1)
2) CO addition
The direct reaction of CO with arynes is interesting as it provides direct access to anthraquinones and fluorenones. These compounds are widely used moieties in various fields, from medicine to material chemistry or organic electronics.[3,4]
The benzyne carbonylation can be catalysed with cheap first-row transition metals such as cobalt[5], which coincidentally also helps with in-situ aryne stabilisation. Using different metal catalysts (Ni[6], Rh[7]) could then alter the reaction selectivity and allow access to different products. (Figure 1) An important part of this step is optimising the reaction conditions starting from the build-up of the catalyst, solvent, temperature and current. In this regard, an intriguing possibility is also an application of UV light on the solution in the presence or absence of CO. This will trigger the rearrangement of benzyne into ketene and open the door to a possibly different reactivity.[8]
Figure 1: Illustration of the forseen aryne carbonylation process
3) Convert CO2 reduction into aryne carbonylation
Subsequently, the combination of CO2 reduction and aryne generation can be done in two ways: through the sequential line-up and one-pot reaction (Figure 2). The CO2 to CO reduction can be achieved either through electrocatalytic cathodic reduction with transition metal catalysts or using contemporary state-of-the art photoreduction using CdS nanonsheets.9 In the case of the separated reactions, the simpler set-up counts with two (electro)chemical cells connected through a tube bridge, allowing CO to cross from the evolution chamber to the aryne solution (Figure 2a). This set-up offers an experimental simplicity; however, it poses challenges to selectivity as CO2 can enter together with CO. However, issues might appear regarding scalability, time efficiency and the reliance of electrochemical reactions on mass transfer. Finally, the more sophisticated approach uses flow chemistry (Figure 3b). In this set-up, it is possible to maximise the efficiency of both electrochemical processes, avoid selectivity issues and decrease the reaction time while quickly scaling up the output.
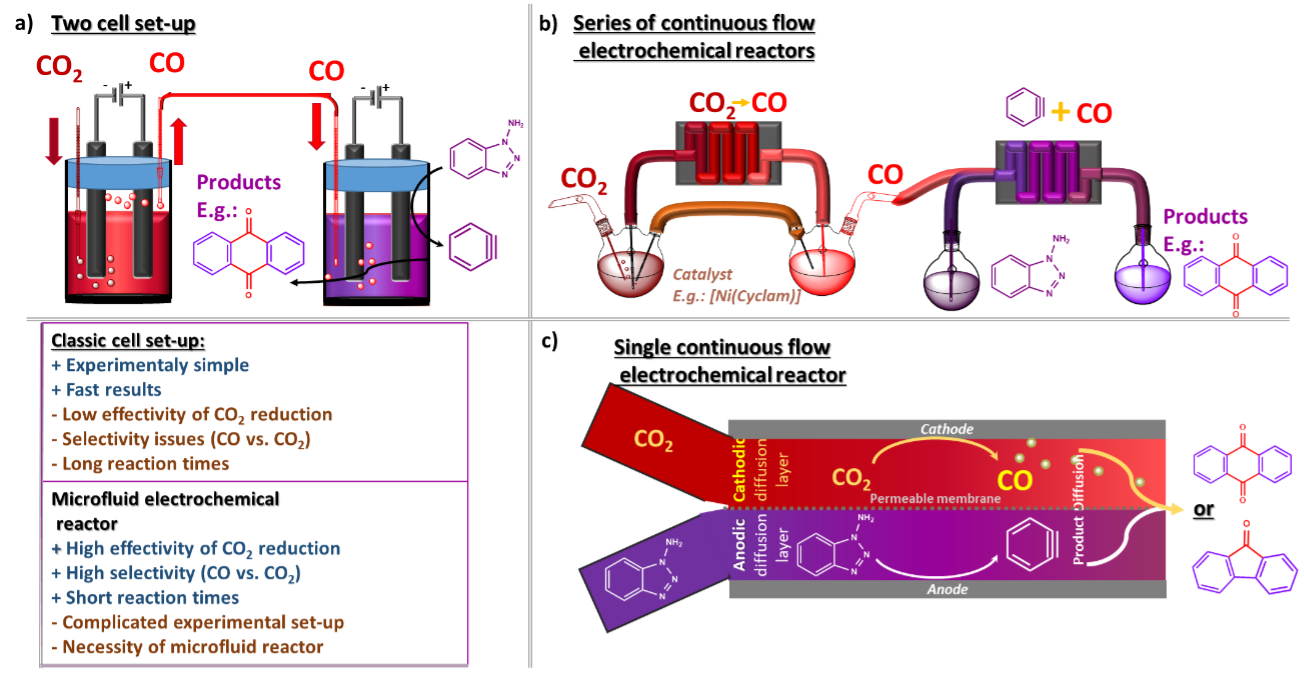
Figure 2: Illustration of different experimental set-ups for effectively coupling two electrochemical reactions into one process. a) Two separate classic electrochemical cells; b) Two microfluidic reactors in series; c) One-pot process in a single microfluidic reactor
The ultimate goal is then to connect both electrochemical processes in one reaction set-up. Again due to the possible competition between CO2 and CO, the prefered approach is to use a chemical flow reactor (Figure 2c). The limited diffusion between the anodic and cathodic layers allows for better effectivity of initial individual transformations, resulting in higher selectivity.[10,11] This is possible simply because, at first, the reactants react in their respective layers. At the same time, the diffusion-conditioned intermolecular reaction happens in distant parts of the reactor where the starting reactants have already transformed into intermediates (e.g. CO2 to CO).
Literature:
[1] M. T. Jensen, M. H. Rønne, A. K. Ravn, R. W. Juhl, D. U. Nielsen, X. M. Hu, S. U. Pedersen, K. Daasbjerg, T. Skrydstrup, Nat. Commun. 2017, 8
[2] L. Li, Y. Li, N. Fu, L. Zhang, S. Luo, Angew. Chemie - Int. Ed. 2020, 59, 14347–14351.
[3] S. Patel, B. Rathod, S. Regu, S. Chak, A. Shard, ChemistrySelect 2020, 5, 10673–10691.
[4] E. M. Malik, C. E. Müller, Med. Res. Rev. 2016, 36, 705–748.
[5] N. Chatani, A. Kamitani, M. Oshita, Y. Fukumoto, S. Murai, J. Am. Chem. Soc. 2001, 123, 12686–12687.
[6] B. L. Edelbach, R. J. Lachicotte, W. D. Jones, Organometallics 1999, 18, 4660–4668.
[7] C. N. Iverson, W. D. Jones, Organometallics 2001, 20, 5745–5750.
[8] J. G. Radziszewski, J. Waluk, P. Kaszynski, J. Spanget-Larsen, J. Phys. Chem. A 2002, 106, 6730–6737.
[9] N. Wang, S. Cheong, D.-E.Yoon, P. Lu, H. Lee, Y. K. Lee, Y.-S. Park, D. C. Lee, J. Am. Chem. Soc. 2022, 144, 16974-16983
[10] T. Noël, Y. Cao, G. Laudadio, Acc. Chem. Res. 2019, 52, 2858–2869.
[11] D. Horii, T. Fuchigami, M. Atobe, J. Am. Chem. Soc. 2007, 129, 11692–11693.







